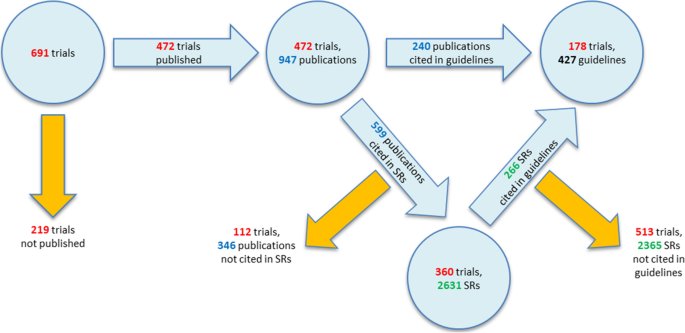
Investigator initiated trials versus industry sponsored trials - translation of randomized controlled trials into clinical practice (IMPACT) | BMC Medical Research Methodology | Full Text

Sponsor-Investigators Frequently Cited in FDA Warning Letters. Do You Know What Is Required for a High Quality Trial? — Clinical Pathways

Industry Viewpoints: Investigator-Initiated Studies – A Challenge for Supply with IMPs? - Clinical Trials Arena

A meta-research study revealed several challenges in obtaining placebos for investigator-initiated drug trials - ScienceDirect

Investigator Initiated Studies, what are they and how can they be of value to your company? - Qserve CRO

Investigator Initiated Trials (IITs) Simplified: A Practical Guide for Clinical Trial Investigators to Conduct IITs: 9781540762894: Medicine & Health Science Books @ Amazon.com