Medicina | Free Full-Text | Effectiveness and Safety of Dabigatran Reversal with Idarucizumab in the Taiwanese Population: A Comparison Based on Eligibility for Inclusion in Clinical Trials
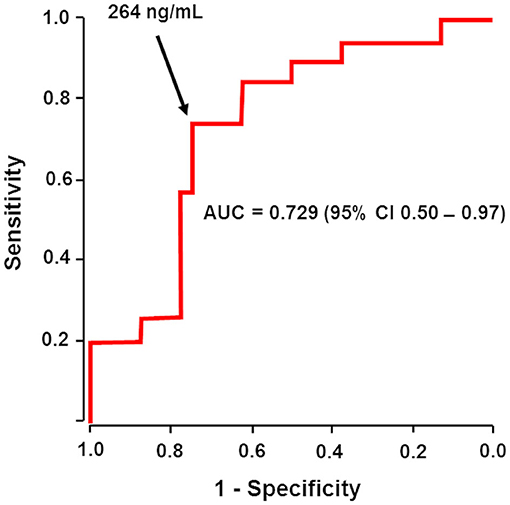
Frontiers | Dabigatran Level Before Reversal Can Predict Hemostatic Effectiveness of Idarucizumab in a Real-World Setting
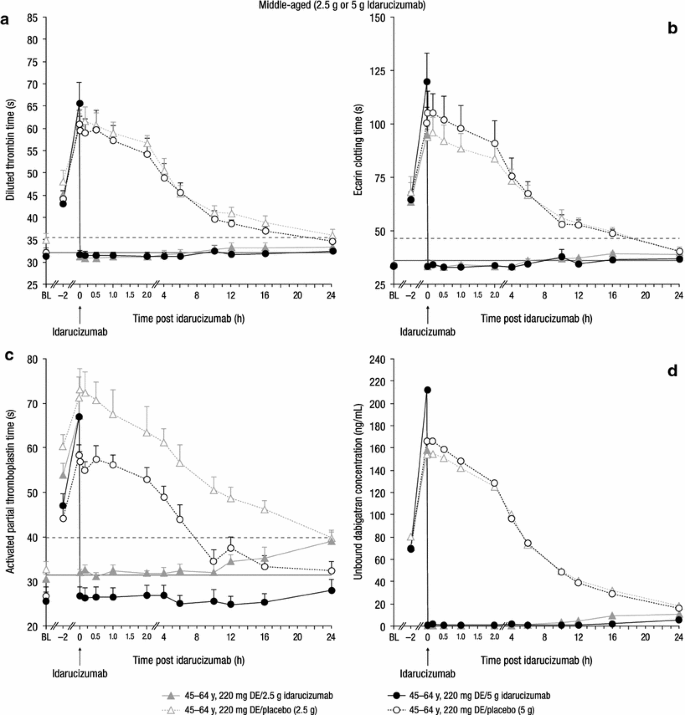
Effect of Age and Renal Function on Idarucizumab Pharmacokinetics and Idarucizumab-Mediated Reversal of Dabigatran Anticoagulant Activity in a Randomized, Double-Blind, Crossover Phase Ib Study | SpringerLink

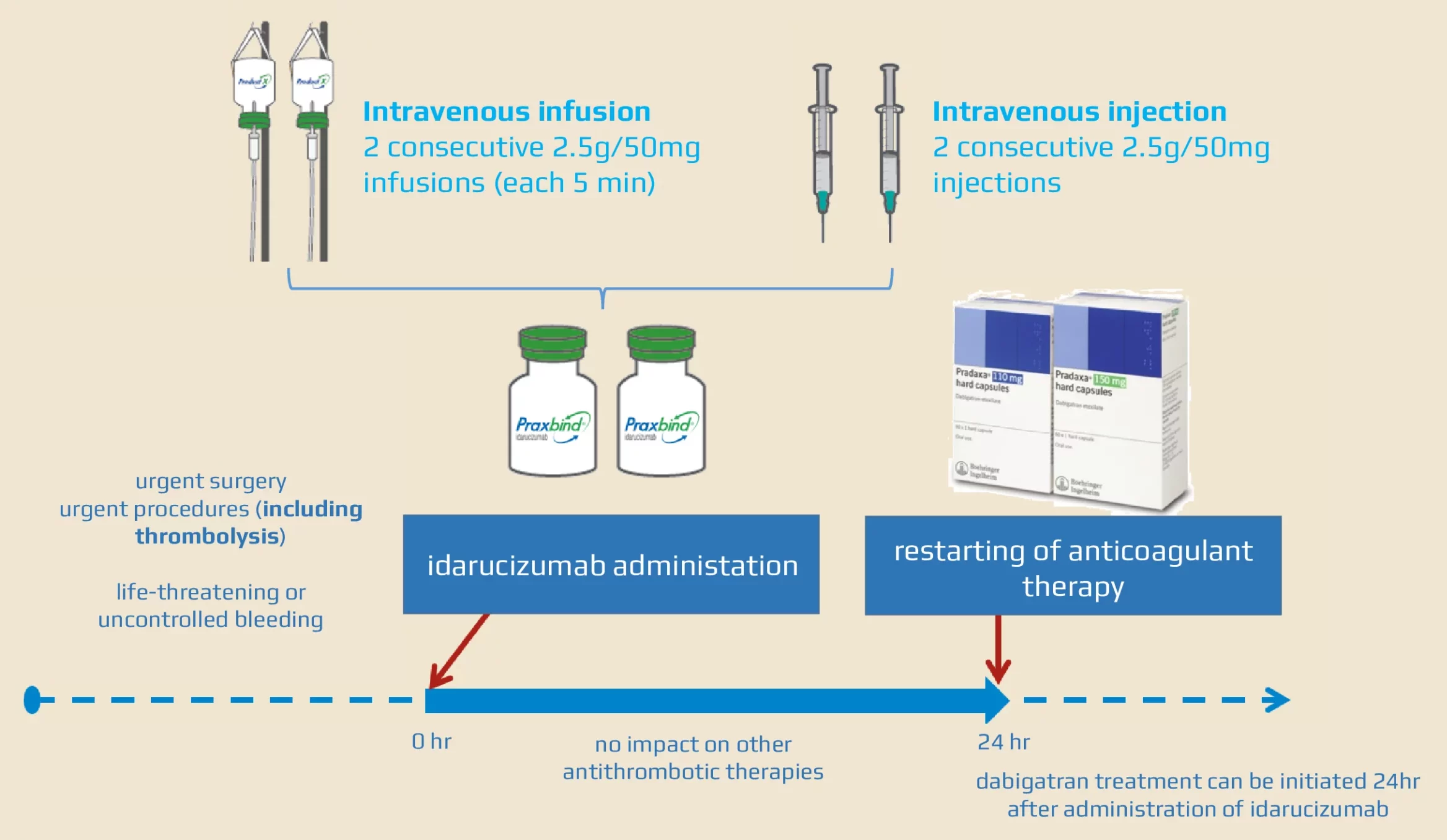
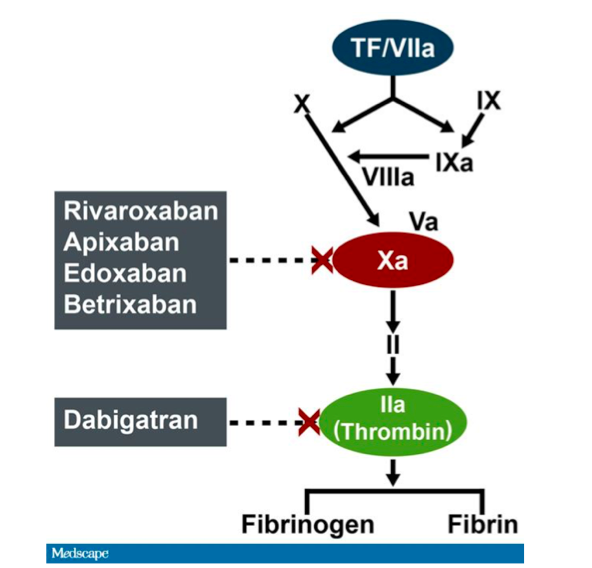
Idarucizumab and factor Xa reversal agents: role in hospital guidelines and protocols - ScienceDirect

Idarucizumab, a Specific Reversal Agent for Dabigatran: Mode of Action, Pharmacokinetics and Pharmacodynamics, and Safety and Efficacy in Phase 1 Subjects - ScienceDirect
Interpretation of idarucizumab clinical trial data based on spontaneous reports of dabigatran adverse effects in the French phar
Idarucizumab, a Specific Reversal Agent for Dabigatran: Mode of Action, Pharmacokinetics and Pharmacodynamics, and Safety and Ef

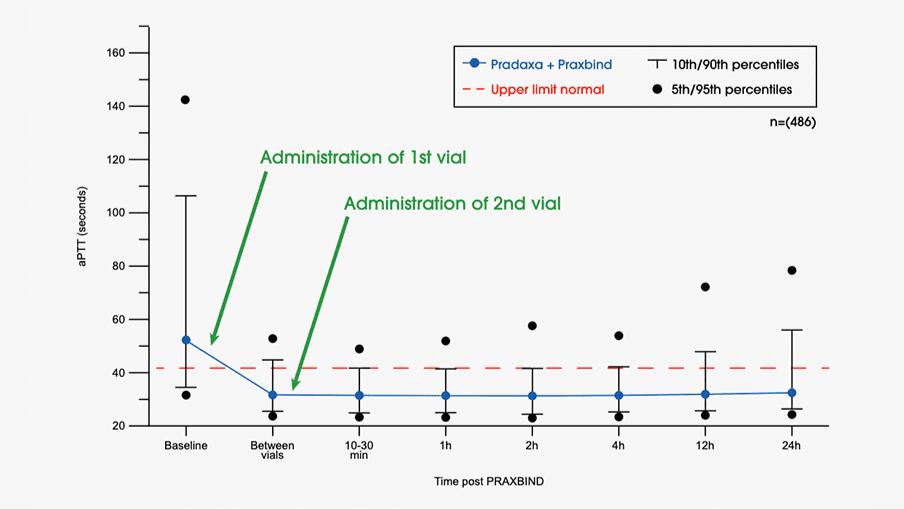
Design of the Reversal Effects of Idarucizumab on Active Dabigatran... | Download Scientific Diagram

Dabigatran Reversal With Idarucizumab in Patients With Renal Impairment | Journal of the American College of Cardiology

Safety, pharmacokinetics and pharmacodynamics of idarucizumab, a specific dabigatran reversal agent in healthy Japanese volunteers: a randomized study - Research and Practice in Thrombosis and Haemostasis

Safety, pharmacokinetics and pharmacodynamics of idarucizumab, a specific dabigatran reversal agent in healthy Japanese volunteers: a randomized study - Yasaka - 2017 - Research and Practice in Thrombosis and Haemostasis - Wiley Online Library

PDF) Effectiveness and Safety of Dabigatran Reversal with Idarucizumab in the Taiwanese Population: A Comparison Based on Eligibility for Inclusion in Clinical Trials

These highlights do not include all the information needed to use PRAXBIND safely and effectively. See full prescribing information for PRAXBIND. PRAXBIND® (idarucizumab) injection, for intravenous useInitial U.S. Approval: 2015
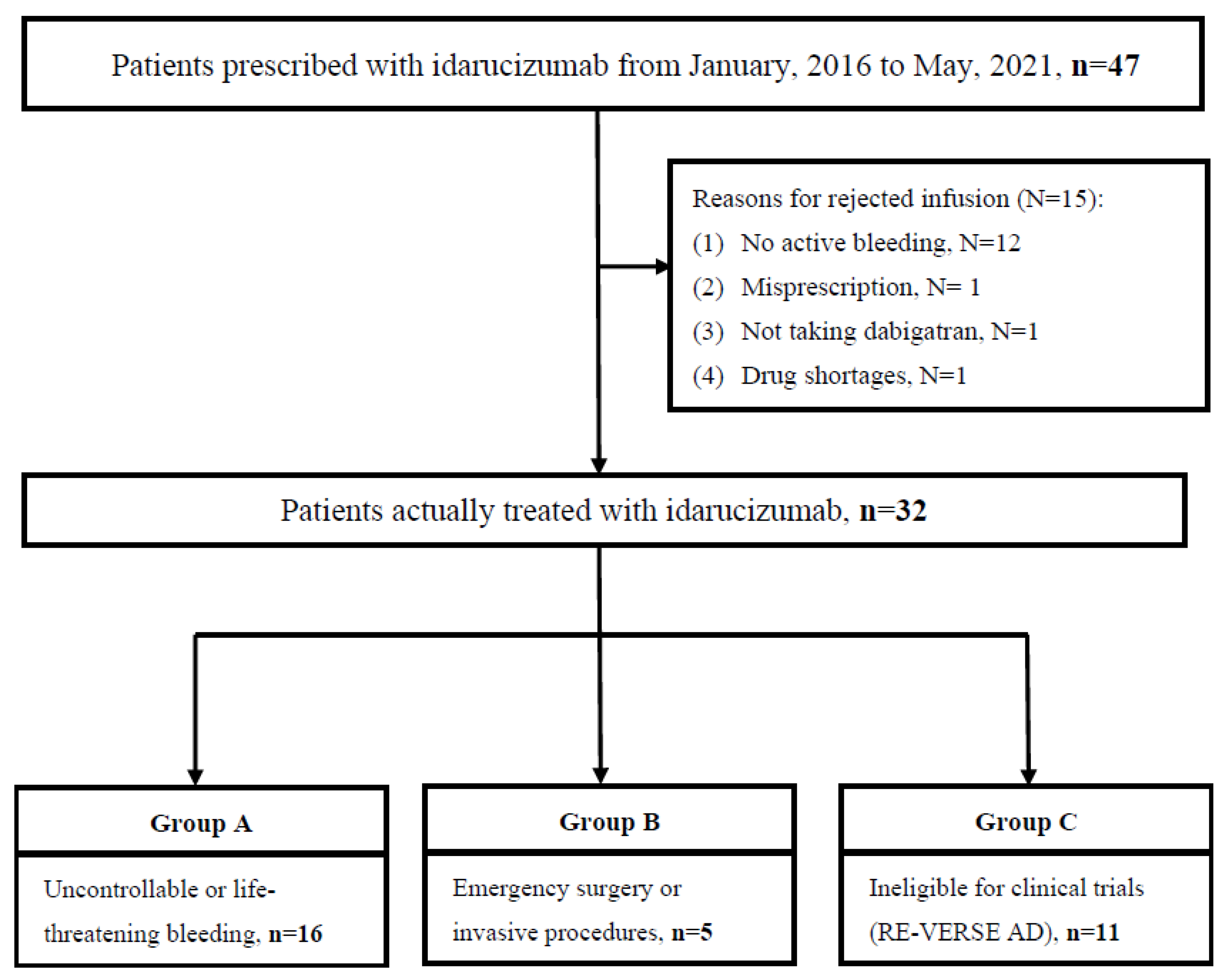
Medicina | Free Full-Text | Effectiveness and Safety of Dabigatran Reversal with Idarucizumab in the Taiwanese Population: A Comparison Based on Eligibility for Inclusion in Clinical Trials
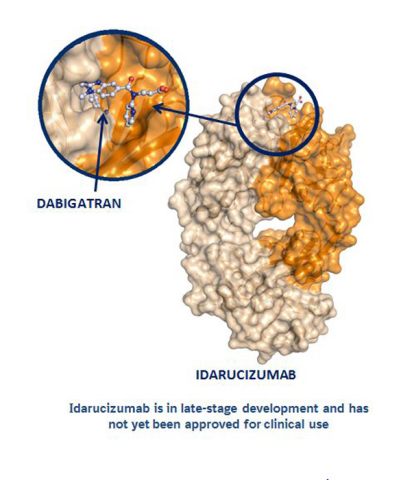
Boehringer Ingelheim submits applications for approval of Idarucizumab, an anti-anticoagulant - Labiotech.eu
Clinical Effectiveness of Idarucizumab in Dabigatran Reversal | Wang | Journal of Cardiology and Therapy
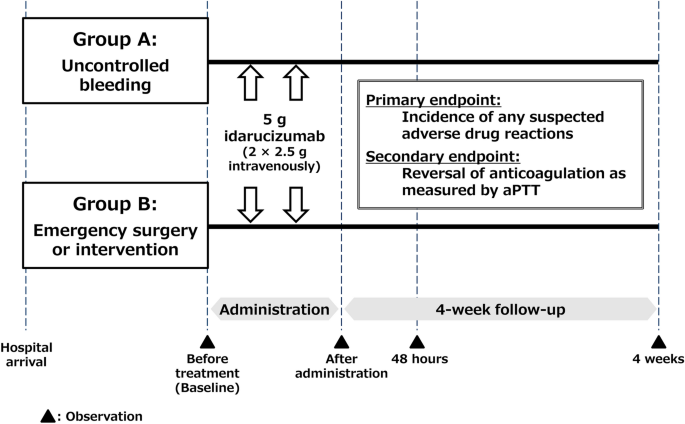
Idarucizumab for Emergency Reversal of Anticoagulant Effects of Dabigatran: Interim Results of a Japanese Post-Marketing Surveillance Study | SpringerLink

Safety, tolerability, and efficacy of idarucizumab for the reversal of the anticoagulant effect of dabigatran in healthy male volunteers: a randomised, placebo-controlled, double-blind phase 1 trial - The Lancet
Usefulness of initial plasma dabigatran concentration to predict rebound after reversal | Haematologica

Idarucizumab for dabigatran reversal: the first 6 months in a tertiary centre - Wheeler - 2019 - Internal Medicine Journal - Wiley Online Library