Directive 2001/20/EC : Clinical trials on medicinal products for human use - Free PDF download | M A N O X B L O G

White Paper: Pharmaceutical Industry Challenges Facing Clinical Trial Disclosure and Transparency - TrialAssure

When innovation outpaces regulations: The legal challenges for direct‐to‐patient supply of investigational medicinal products - Malone - 2022 - British Journal of Clinical Pharmacology - Wiley Online Library

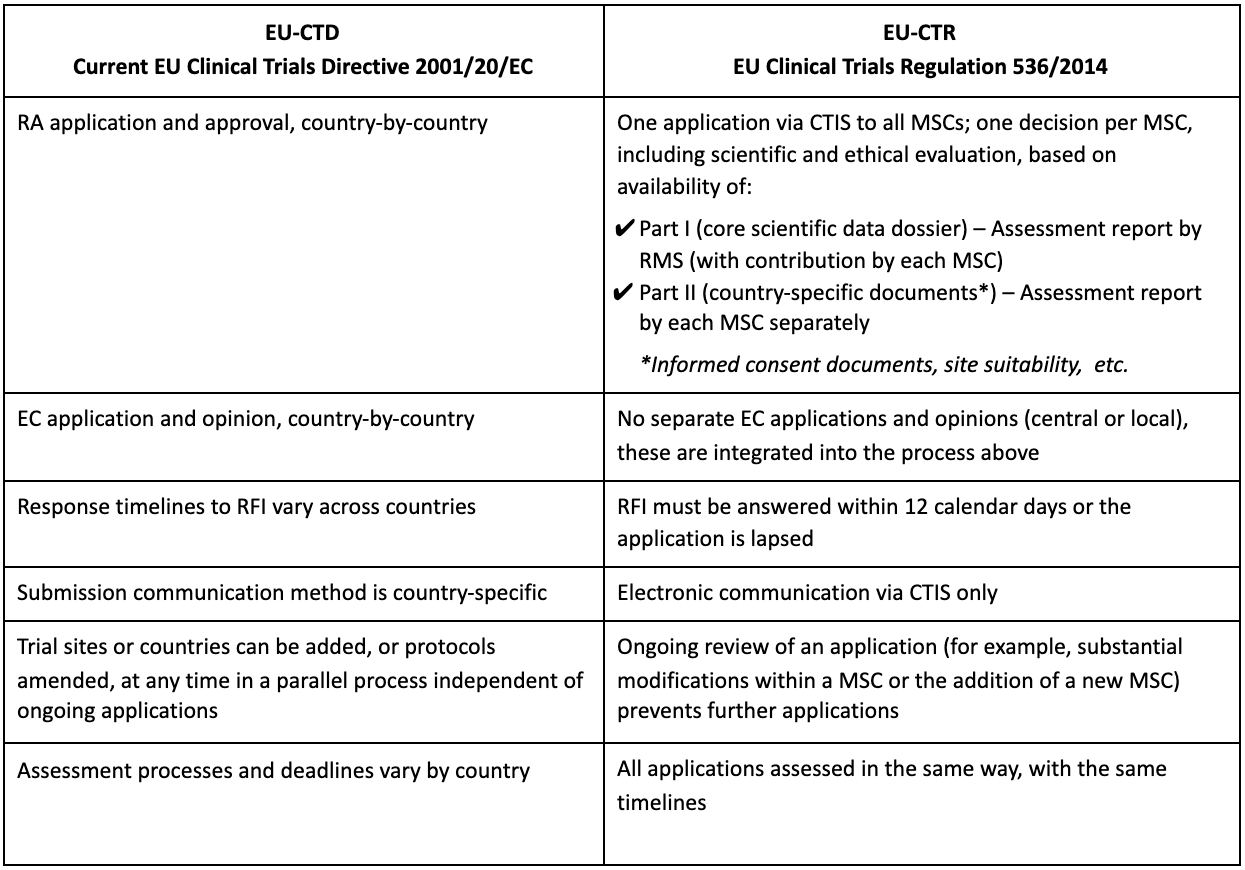
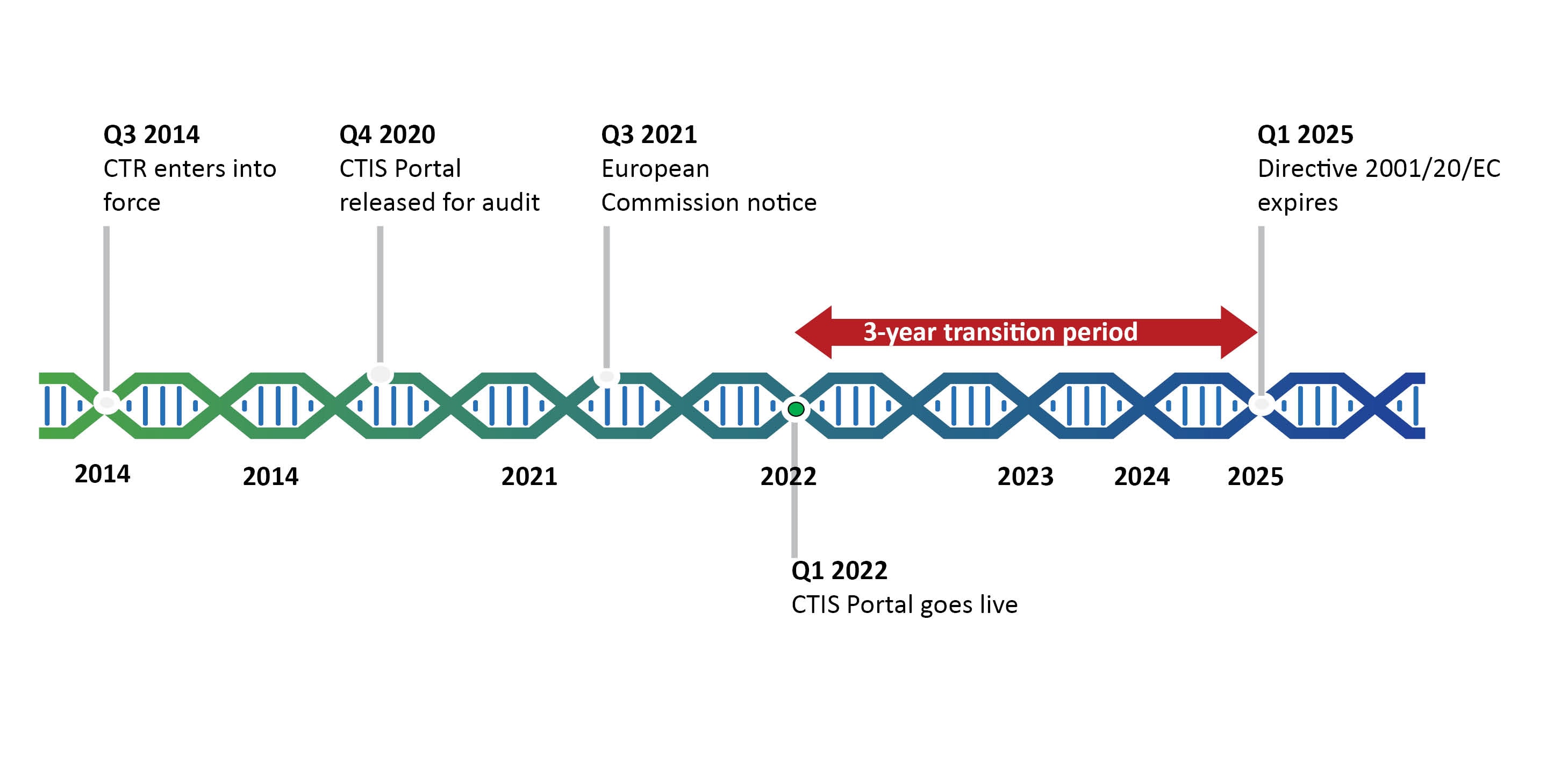
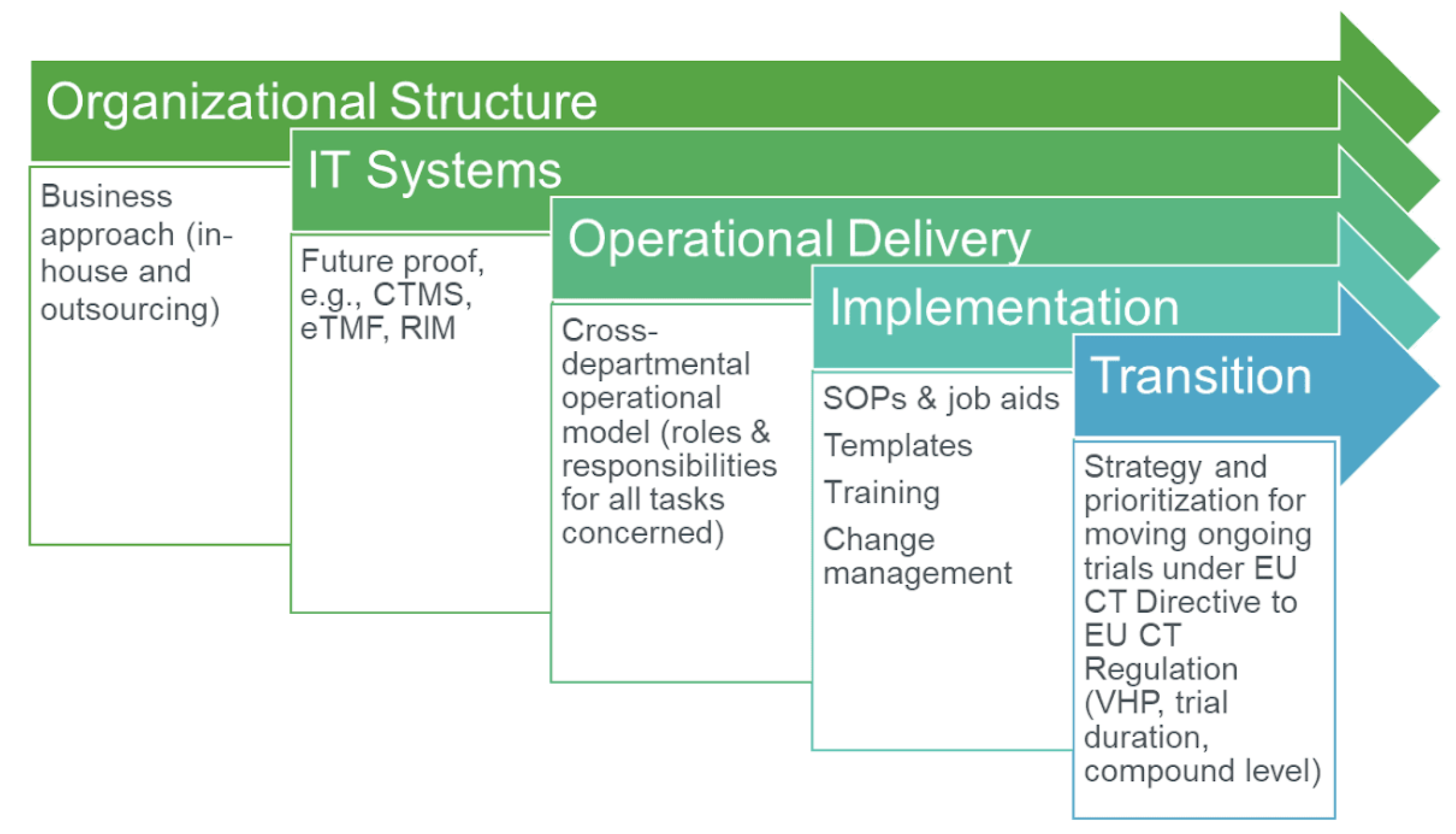
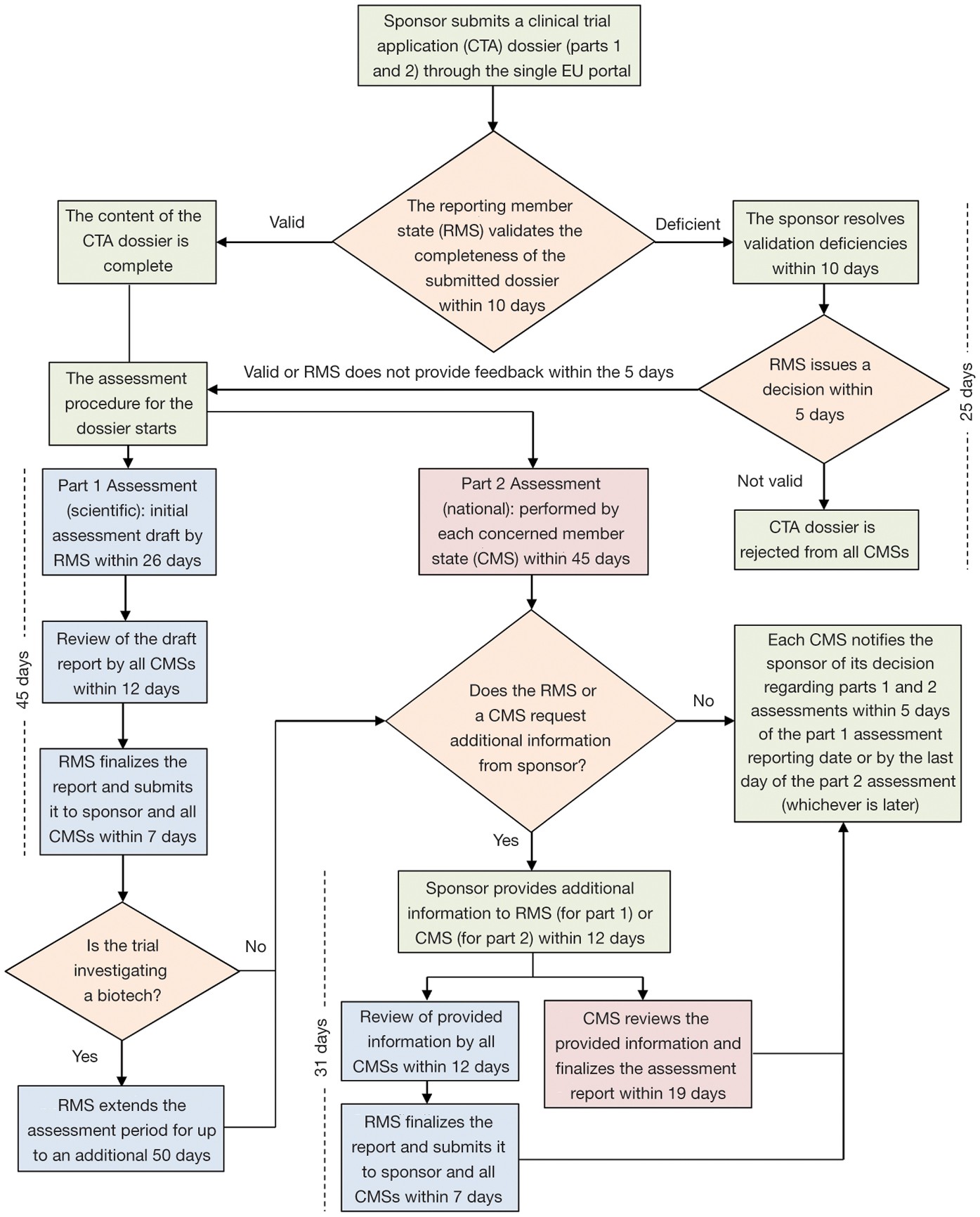
GCP and Quality in “Regulation (EU) 536/2014 on clinical trials on medicinal products for human use and repealing Directive 2001/20/EU” - ScienceDirect
EUROPEAN COMMISSION Brussels, 11/04/2012 sanco.ddg1.d.6(2012)501417 VOLUME 10 - G Date of discussion of draft by the ad-hoc gro

PDF) Typical investigational medicinal products follow relatively uniform regulations in 10 European Clinical Research Infrastructures Network (ECRIN) countries