Randomized Trials Fit for the 21st Century: A Joint Opinion From the European Society of Cardiology, American Heart Association, American College of Cardiology, and the World Heart Federation | Journal of the

Clinical Trial Guidelines Ppt Powerpoint Presentation Inspiration Example Introduction | PowerPoint Slides Diagrams | Themes for PPT | Presentations Graphic Ideas
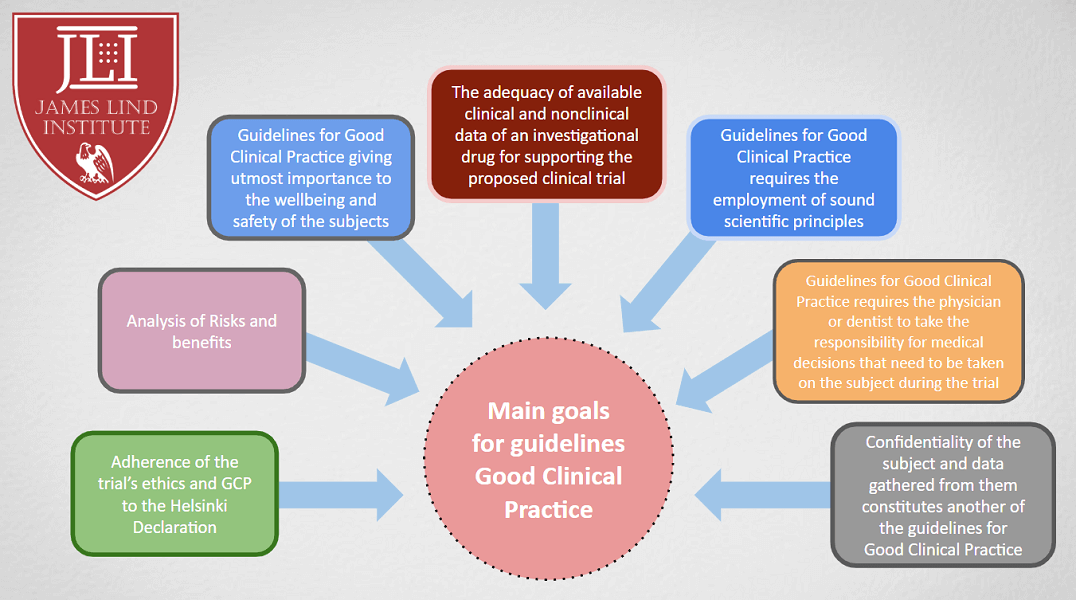
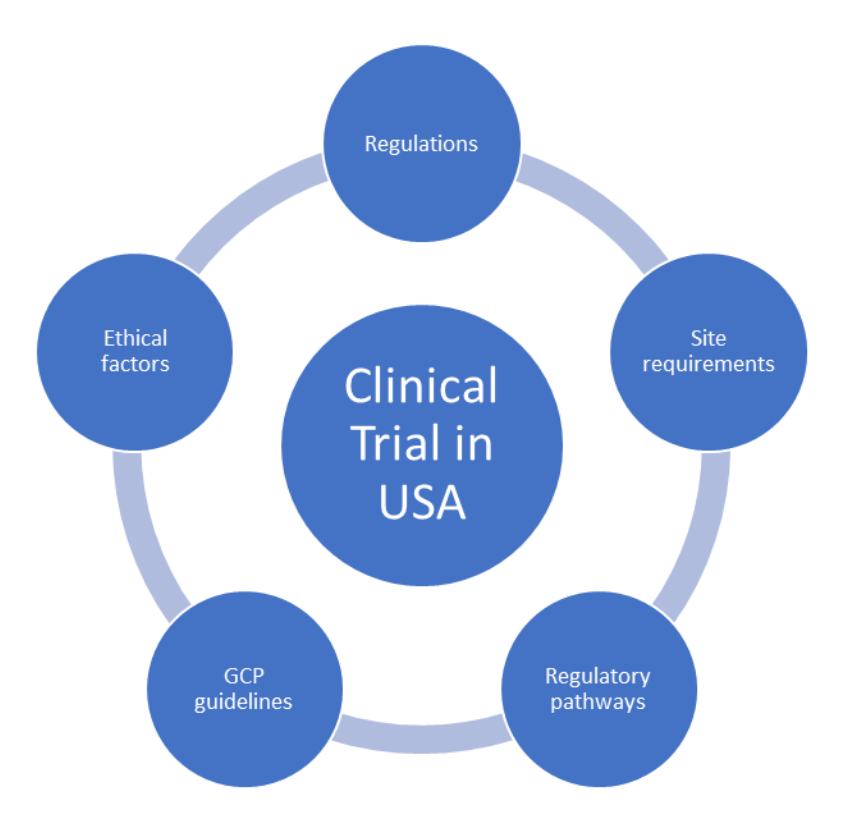
![PDF] RECENT ADVANCEMENT IN REGULATORY GUIDELINES FOR CLINICAL TRIALS IN USA AND INDIA | Semantic Scholar PDF] RECENT ADVANCEMENT IN REGULATORY GUIDELINES FOR CLINICAL TRIALS IN USA AND INDIA | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/52893bcfc57fecda0c60e34dd1fe46414c867575/9-Figure4-1.png)
PDF] RECENT ADVANCEMENT IN REGULATORY GUIDELINES FOR CLINICAL TRIALS IN USA AND INDIA | Semantic Scholar

Updated clinical trial reporting guidelines include patient voices to improve trial utility and transparency

Early phase oncology clinical trials in Malaysia: current status and future perspectives - Voon - 2023 - Asia-Pacific Journal of Clinical Oncology - Wiley Online Library

When innovation outpaces regulations: The legal challenges for direct‐to‐patient supply of investigational medicinal products - Malone - 2022 - British Journal of Clinical Pharmacology - Wiley Online Library
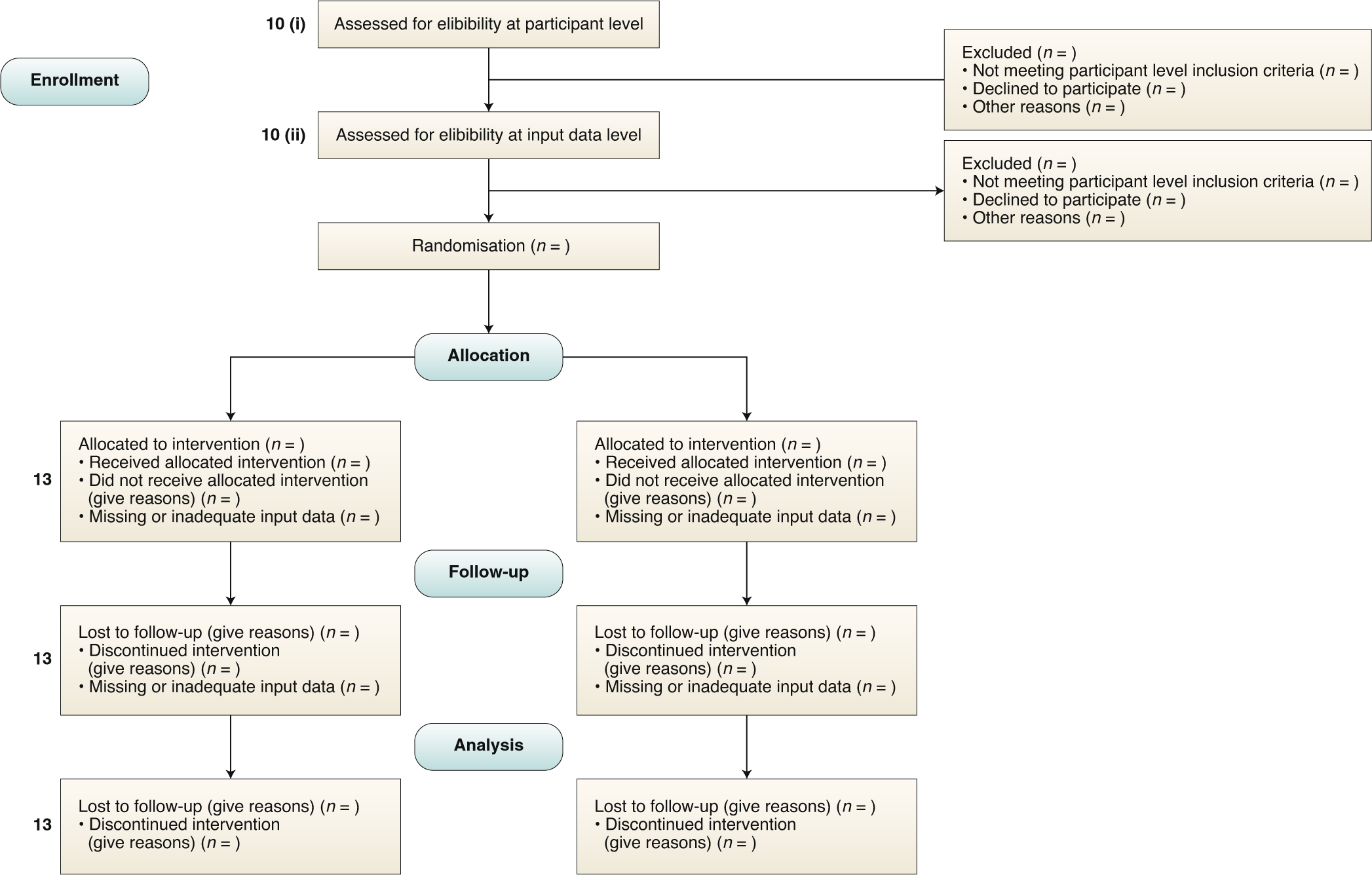
Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension | Nature Medicine

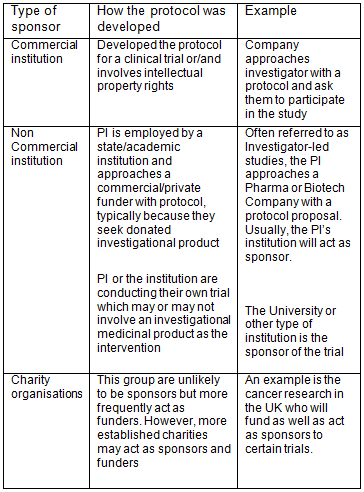
Comparison of clinical trial guidelines in USA, EU and India, Singapore. | Download Scientific Diagram
![PDF] RECENT ADVANCEMENT IN REGULATORY GUIDELINES FOR CLINICAL TRIALS IN USA AND INDIA | Semantic Scholar PDF] RECENT ADVANCEMENT IN REGULATORY GUIDELINES FOR CLINICAL TRIALS IN USA AND INDIA | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/52893bcfc57fecda0c60e34dd1fe46414c867575/14-Table2-1.png)
PDF] RECENT ADVANCEMENT IN REGULATORY GUIDELINES FOR CLINICAL TRIALS IN USA AND INDIA | Semantic Scholar